Ingestible Biosensors
An electronic "pill" could optimize physiological status monitoring.
Daytime summer temperatures in Iraq and Afghanistan easily soar into the triple digits. Troops stationed in such hostile environments face a dry, desert-like climate, all the while lugging around heavy gear and equipment. Not surprisingly, they are at a high risk for heat exhaustion, heat stroke, and serious heat-induced complications, such as liver damage and kidney failure.
Knowing a soldier’s core temperature—as well as heart rate and breathing rate—can lead not only to risk mitigation and preventive medicine, but also to operational performance enhancement and post-combat injury monitoring. Physiological status monitoring (PSM) systems, such as heart rate chest straps and ingestible core temperature pills, are already on the market; however, these devices suffer from limited operational utility because of uncomfortable design elements, insufficient battery life, poor signal reliability during an individual’s movement, and inability to measure multiple physiological parameters at once and with a single device.
The U.S. Army Research Institute of Environmental Medicine (USARIEM) is constantly seeking better ways to monitor human physiology, according to Albert Swiston, a biomaterials scientist in the Bioengineering Systems and Technologies Group at Lincoln Laboratory. Swiston, along with fellow group members Tadd Hughes and Gregory Ciccarelli, and MIT collaborators Giovanni Traverso and Professor Robert Langer (Department of Chemical Engineering), are working toward this USARIEM goal. Together, they have been developing a single ingestible device capable of simultaneously measuring core temperature, heart rate, and breathing rate.
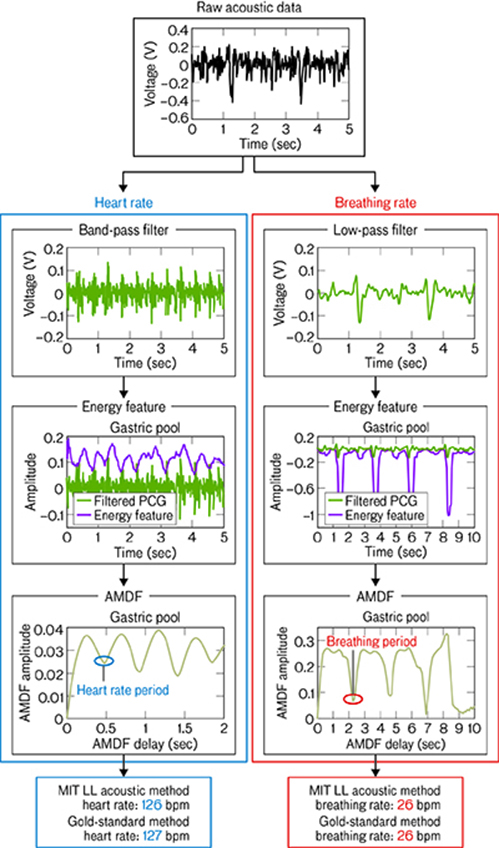
The device will feature two sensors: a thermistor and a microphone. The thermistor senses temperature variations based on changes in electrical resistance. The microphone, or “digital stethoscope” as Swiston describes it, is sensitive to the frequencies of acoustic signals from the heart and lungs. Typically, these cardiac and respiratory vibrations are converted into a graphical representation known as a phonocardiogram. In this device, however, signals will be fed through an amplifier and a series of analog filters. These filtered data will then be sent to a microcontroller that performs simple digital-signal processing to determine heart rate and breathing rate.
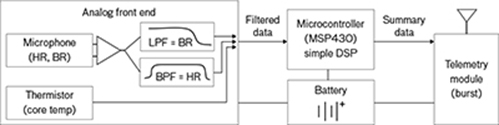
Schematic of an ingestible electronic device that concurrently measures heart rate (HR), breathing rate (BR), and core temperature (CT). Acoustic signals recorded from the microphone are fed through a low-pass filter (LPF) and band-pass filter (BPF) and then sent to a microcontroller that performs simple digital-signal processing (DSP) to determine HR and BR.
So far, the system has only been modeled on pigs. With an almost identical gastrointestinal (GI) tract, an equivalent heart and lung size, and similar body weight to a full-grown human, a pig closely mimics human physiology. To test the system, an endoscope (a tubular internal viewing instrument) guided a microphone through every point along a pig’s GI tract, where acoustic data were recorded. External vital signs were also taken to compare to the acoustic data derived from the team’s internal microphone-based method. The results show a high degree of accuracy between the datasets for both heart rate and breathing rate. (This model did not test the thermistor component of the system because thermistors are already widely accepted to be one of the most accurate types of temperature sensors.)
Swiston says the team “would really like to get to the point of having the pig swallow a device.” With current commercial components, the team estimates the first prototype will be 2 cm in length and 1 cm in diameter, but the team is confident that the device can be further reduced in size to facilitate ingestion. “This device will be smaller than many vitamins people voluntarily take on a daily basis,” comments Swiston.
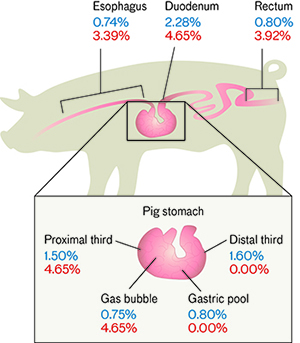
The percentage difference between externally derived HR and BR data using gold-standard methods and internally derived HR and BR data from a phonocardiogram/average magnitude difference function signal processing analysis as recorded by an endoscopically guided microphone at all sites along a pig’s gastrointestinal tract. For each site, note the low percent error rates for both HR (blue) and BR (red).
According to Swiston, the team is on the “cusp of building an actual product” with unit parts estimated to be under $10. The device will need to be made of a material resistant to acid, perhaps a polycarbonate sheathing. “This decision will be made later when we know exactly how big the electronics are,” Swiston continues. Another decision will involve power consumption, a requirement that will depend on whether the device is intended to be persistent (affixed in the GI tract for some extended period of time) or transient (naturally passed through the GI tract). This consideration is part of a larger concept-of-operations assessment that will need to evaluate several questions:
- What does the user want to know? Does the user want to know heart rate, breathing rate, and core temperature all at once? Does the user only want to know breathing rate? Or does the user want to know a different vital sign altogether?
- For how long does the user need to know this information? Is the user monitoring physiology over the long term or in emergency situations? How frequently do vital signs need to be updated and reported?
- Can the person ingesting the device tolerate it? Would GI-related complications or other medical conditions inhibit the proper functioning of the device?
For now, the team will focus on building a transient, ingestible PSM pill that can last up to 24 hours and simultaneously measure heart rate, breathing rate, and core temperature.
The finalized device will have broad applicability not only in warfighter health and performance but also in sports and medicine. “Performance athletes want to know their heart and breathing rates with high fidelity,” Swiston notes. “Marathon runners or National Football League players can swallow the pill right before a race or game or training practice.” Swiston continues, “This device would also be great for burn victims. Measuring their vital signs is extremely important, but putting electrocardiogram (ECG) electrodes on their already fragile skin is, of course, unpleasant.” Other applications of the device include triaging patients in the emergency room, monitoring the live-at-home elderly, warning surgical patients of complications like postoperative fever, and detecting cardiac murmurs and asthma, for instance.
Schematic of an ingestible electronic device that concurrently measures heart rate (HR), breathing rate (BR), and core temperature (CT). Acoustic signals recorded from the microphone are fed through a low-pass filter (LPF) and band-pass filter (BPF) and then sent to a microcontroller that performs simple digital-signal processing (DSP) to determine HR and BR.
Swiston envisions the device will eventually be able to measure other important physiological parameters, such as the saturation of oxygen in the blood and pH levels. He also foresees the evolution of an “ingestible modular device with individually ‘prescribed’ components most appropriate for a patient’s medical condition” to address the different needs of military and civilian users. Another possibility involves using a persistent PSM pill as a drug-delivery mechanism that would be induced by specific changes in an individual’s physiological status. A similar system may be useful for delivering powerfully therapeutic bacteria, particularly anaerobic bacteria (bacteria that live and grow only in the absence of oxygen), directly into the GI tract.
Are ingestible electronics the wave of the future? Swiston thinks it is a matter of redefining our cultural norms: “At first, swallowing electronics may be just as novel as brushing your teeth once was. But, who knows, one day we could be swallowing these devices every day!” With the expanding field of ingestible electronics—backed by the now decade-long Food and Drug Administration approval of their clinical use—this idea may be an easy pill to swallow after all.
|